|
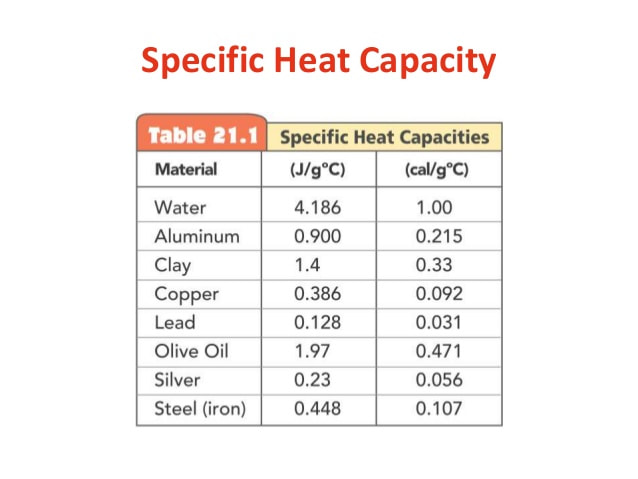
Today, we learned about the concept of "Specific Heat Capacity".
What I learned: - What Specific Heat Capacity is
- What a calorie is
- What intrigued me:
- Question I had:
0 Comments
Today, we conducted experiments to gain a better understanding of thermal energy transfers. One experiment required us to predict whether a plastic or metal object was colder and the other required us to predict which balloon (one with water inside, one without water inside) would pop first when heated.
What I learned: - Why some objects feel colder than others when they're the same temperature?
What intrigued me:
Questions I had:
Reflection:
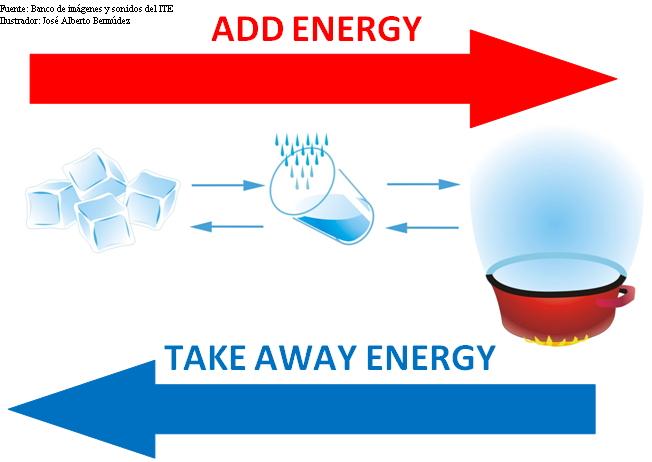
Today, we learned the concept of "Internal Energy" (thermal energy). We did an experiment with lukewarm water and boiled water and observed which one spread food colouring faster. The boiled water did.
What I learned: - What internal energy is
- What thermal energy is
- Atoms and molecules are always in constant motion
- Temperature determines the direction of flow of thermal energy between bodies
- How to change the internal energy of a substance.
- Gravitational potential energy is stored between atoms
- Two objects will always reach thermal equilibrium if left alone, through conduction or convection. - Using a Kelvin as a unit of temperature
What intrigued me:
Questions I had:
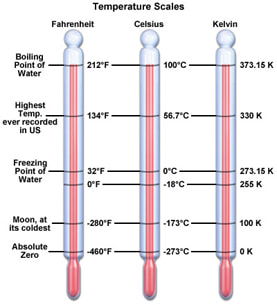
Today, I took notes on a powerpoint about thermal physics and chose specifically to focus on temperature.
What I learned: - The definition of temperature
- Ways to measure temperature
- The Kelvin Scale
- Thermal Energy depends on
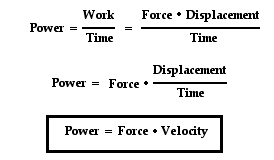
Today, we were introduced to the concept of "Power". We were to do an experiment requiring us to measure our mass, and the height of a set of stairs and how long it took us to climb up, in order to measure the power used.
What I learned: - What "power" is
- LOL diagrams are a much better way to determine work done in comparison to free-body diagrams. - What intrigued me:
- Questions I had:
- Reflection
Today, we worked on our lab. We were to drop a marble down an incline and measure the height it was dropped at and the exit velocity of the marble. We would repeat this process 5 times and 5 different heights and record the results. This would leave us with 25 data points we could use to put in a chart and later turn into a graph,
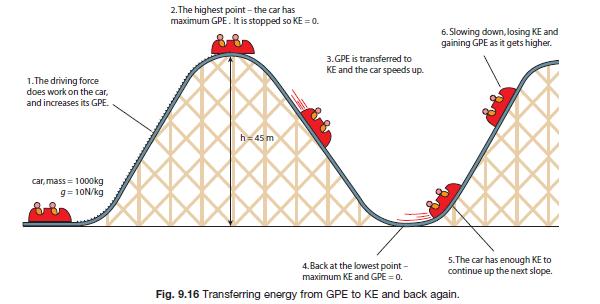
What I learned: - How to use the LabQuest to record the instantaneous exit velocity of an object. - The independent variable was the height. - The dependent variable was the exit/final velocity. What intrigued me: - The idea that this technology is similar to what is used to professional sports like running, tennis and baseball to record the speed of an object. Today, we were introduced to the concept of "Conservation of Energy". We worked on a worksheet that was about finding the velocity of a roller coaster.
What I learned: - The concept of Conservation of Energy
Questions I had:
Reflection:
Today, we finished the gravitational potential energy worksheet and conducted a new experiment. The experiment consisted of two balls (equal in mass) being dropped on different inclines (one steeper than the other). We were to make predictions as to which ball we thought would reach the end point first and what the velocities would be at the end point. After this, we switched one of the balls with a ball that was greater in mass and did the experiment again.
What I learned:
- What intrigued me:
- Reflection
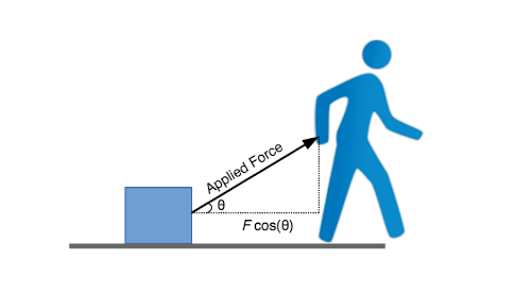
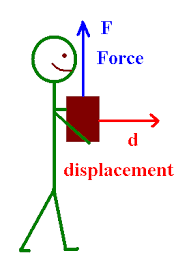
Today, we finished the work worksheet and were introduced to a equation for work.
What I learned: - A new equation for work; W = Fdcosθ
- What intrigued me:
Questions I had:
Today, we continued with our introduction to energy and completed the worksheet we were working on previously. We also were introduced to the concept of "work".
What I Learned: - How to properly draw energy-flow diagrams
- There are 9 different types of energy
- Energy cannot be created or destroyed
- What "work" is
What intrigued me:
Questions I had:
Reflection:
|
Categories |